|
1 shows that a 12x12x12 grid in k-space is sufficient for an accuracy of 0.005 eV. Moreover, lowest lattice constant requires the highest number of k points and hence 346 pm was chosen for optimizing the number of k points. Since 351 pm is the lattice constant of BCC at room temperature, varying the lattice constant from 346 pm to 356 pm would be sufficient to get an optimum lattice constant at which the energy is minimized. BCC lattice was chosen because at room temperature lithium crystallizes as BCC with a lattice constant of 351 pm. The number of k points was optimized for BCC lattice with a lattice constant of 346 pm. However, before varying the lattice parameter for a given crystal structure, the optimum number of k points and the cutoff energy to be used in calculations were obtained. Minimum energy of different crystal structures was compared to obtain the optimum crystal structure and the corresponding lattice parameter. A core radius of 1 Bohr and an electronic configuration of 1s2 2s1 was used for calculations. For all calculations Generalized Gradient Approximation (GGA) and Perdew-Burke-Ernzerhof (PBE) exchange correlation functionals were used with On The Fly Generated (OTFG) ultrasoft pseudopotential. In order to study the crystal structure of lithium, energy was minimized for different crystal structures by using CASTEP in Material studio. Moreover, repeating calculations in a software is also much cheaper than repeating an experiment. Density Functional Theory (DFT) was used for studying the crystal structure as it is much cheaper than using experimental techniques such as X-ray diffraction. Hence, lithium is one of the most important metals and so the crystal structure of lithium was studied in this project. and lithium metal batteries are amongst the most promising future energy storage technologies. 
Today, lithium ion batteries are being used in variety of applications such as electric vehicles, laptops, mobile phones, grid storage etc. Lithium is a very important component of batteries today. However, SCC has a significantly higher energy compared to BCC and FCC. A comparison of 0 K lattice energies at the optimum lattice parameter suggests that the energy difference between BCC and FCC is lower than 0.005 eV, with BCC having lower energy. Optimum lattice parameters have been obtained for BCC, SCC, and FCC lattices by using plane-wave basis set density functional theory methods. A few examples are given in the link.ĥ.4.1 Partial Dislocations and Stacking FaultsĮxercise 3.In this project, ‘Material Studio’ has been used to study the crystal structure of lithium by using Density Functional Theory (DFT). "diamond type, Perovskites, "Zinkblende" structure and so on. Wigner-Seitz cell and its use in constructing the lattice are shown in yellow.Ĭrystal types instead of lattices by using the name of prominent crystals, crystallographers or minerals etc. Lattice point to neighboring points at half the distance with planes at right angles to the linesīelow: The blue lines connect lattice points, the red lines denote the intersection at right angles. Other cases (especially if working in reciprocal lattices) the choice of a Wigner - Seitz cell may be appropriate, which is obtained by intersecting all lines from one there are unit cells with a smaller volume (but without the cubic symmetry). Whereas, for example, it shows best the cubic symmetry of the cubic lattices, itsĬell of the lattice, i.e. up to huge complex molecules as in the case of The base can just be one atom (as in the case of manyĮlemental crystals, most noteworthy the metals), two identical atoms (e.g. 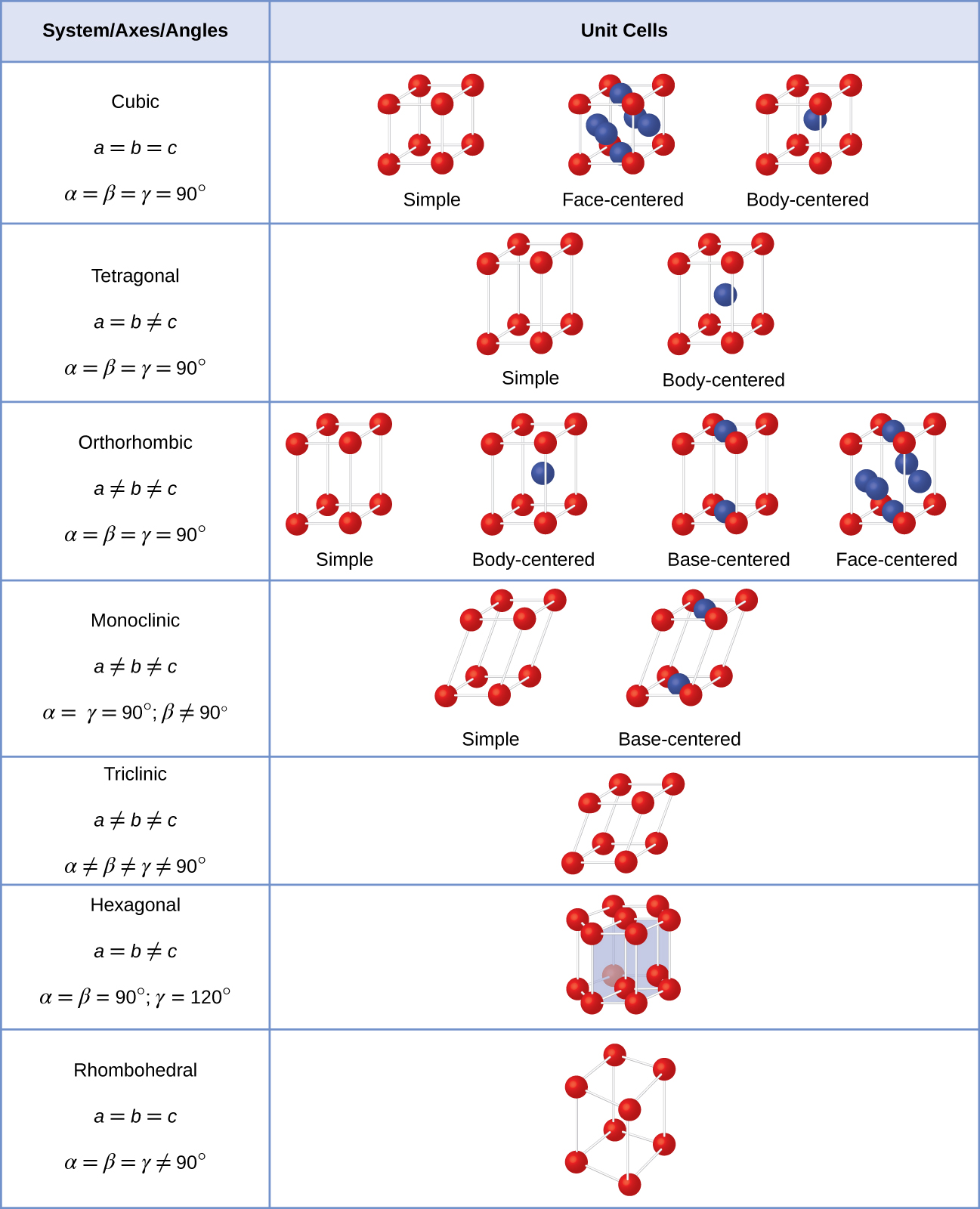
symmetry)Ī crystal now is obtained by taking a Bravais lattice and adding a base! 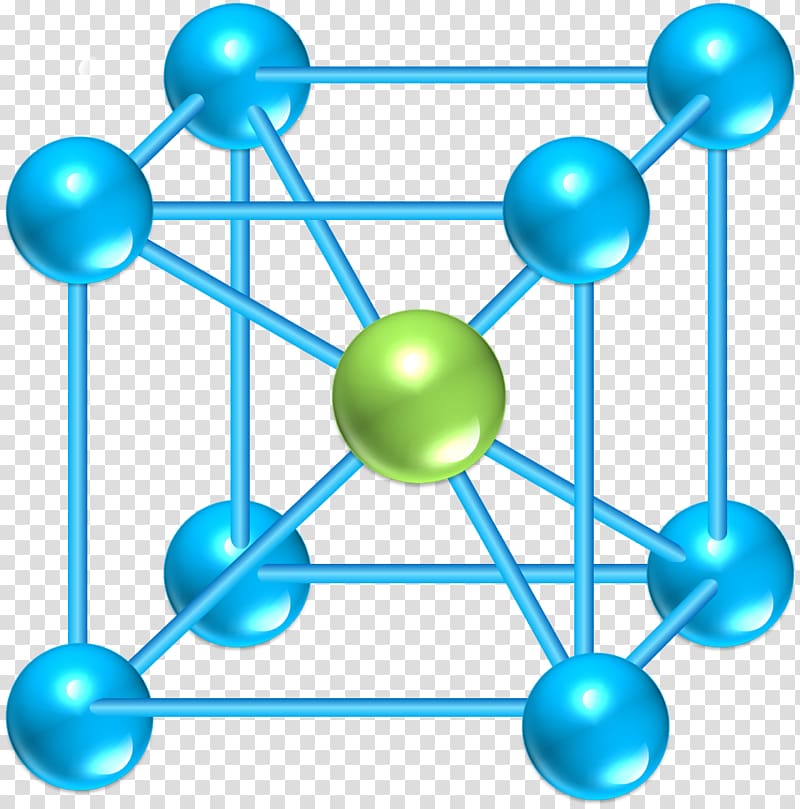
Hexagonal (elementary cell continued to show hex. Points are shown as little spheres and occasionally only "visible" lattice points are shown. Their basic features are shown below (For sake of clarity, the lattice This yields the 14 Bravais lattices, whichĪre commonly used to describe lattice types. Each lattice point than can be reached by a translation vector T ofĬonvenient, to classify lattices according to some basic symmetry groups. Of elemental crystals where the base consists of one atom only.Īll possible lattices can be described by a set of three linearly independent vectors Note that a lattice is notĪ crystal, even so the two words are often used synonymously in colloquial language, especially in the case Structural units (or base of the crystal) are the atoms in some specificĪrrangement which are unambiguously placed at every lattice point. The periodicity is described by a mathematical lattice (which are mathematical points at specific coordinates in space), the identical Identical structural units in three dimensional space.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
